Home / Products / PharmaTainer™ Bottle Solutions / PharmaTainer™ Bottles
Bottle Solutions
PharmaTainer™
Bottles
PharmaTainer™ is a range of PETG, PET and PC bottles and carboys from 10ml to 20L* for biomanufacturing liquid transfer, storage, and transport. All our bottles and caps are produced in an ISO Class 5 (Class 100) environment with 100% virgin ACDF-approved medical-grade resins.
The SaniSure® PharmaTainer™ bottles are certified sterile SAL10-6, and every batch is tested and certified to meet USP788 (particles) and USP85 (endotoxins).
*PETG are 10mL to 10L

Overview
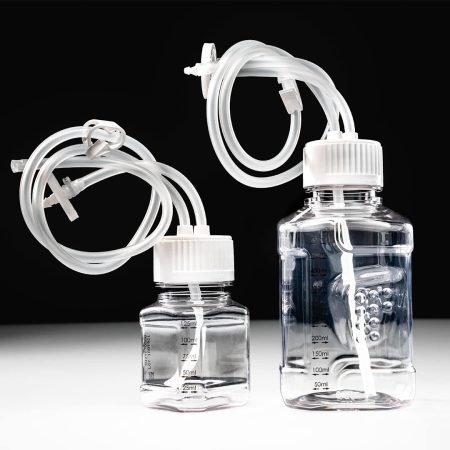
PharmaTainer™ Bottles
Applications
- Media preparation
- Sampling and aliquoting
- Mixing and filling
- Storage at low temperature
- Shipping
Benefits
- From 10ml to 20L in PET or PC
- From 10mL to 10L in PETG square bottles and carboys
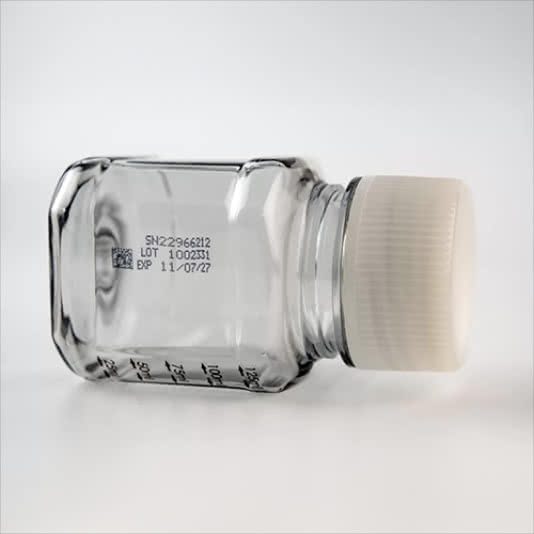
- Traceability with a unique serial number
- Printed graduations with a 2% accuracy
- Caps with perfect leak-proof without a gasket
- Triple packaging aiding delivery into your cleanrooms
Specifications
- Resins ADCF, grade medical with a DMF.
- Certified to meet USP788 for particulate.
- Endotoxins tested according USP 85.
- Certified Sterile SAL 10-6 according a ISO 11137 sterility program.
- Complete file validation with E&L and Biocompatibility studies (ISO 10993-6,-10,-11).
- USP class VI and 21CFR compliant.
Top-quality single-use rigid containers
Traceable
Serial number to allow the bottle to be traced in each step of the process.

Easy to manipulate
The PharmaTainer™ design includes a grip ensuring you can manipulate the bottle without difficulty.

Compliant
Our manufacturing process is ISO 9001-2015 certified, and our cleanrooms are ISO 14644 Class 7 compliant.

No leakage
Our caps are manufactured with a double seal to provide a fail-safe closure. No gasket is required, and no overwinding is possible.
More in PharmaTainer™ Bottle Solutions
Incorporate accessories to your PharmaTainer™
PharmaTainer™ Bottle Solutions can be combined with accessories, like 2-, 3-, and 4-ported caps with hose barbs, closure/opening chucks, and a range of sterile and individual replacement caps in HDPE.
Reach out to us for superior product quality and customization.