Streamlining Biopharmaceutical Solution Preparation & Distribution: A Closed, Single-Use Workflow from Mixing to Sterile Aliquoting
Learn how a closed, single-use workflow eliminates inefficiencies in solution preparation, reduces batch cycle time by over 90%, minimizes product loss, and enhances sterility across mixing and sterile aliquoting processes.

Abstract
Conventional, manual biopharmaceutical solution preparation and distribution workflows introduce significant process risk at every intermediate step — from post-mixing transfers into hold vessels to sequential manual bottle filling and redundant filter integrity tests (FITs).
For bioprocessing engineers managing buffer, media, and process intermediate preparation, these inefficiencies translate directly into extended batch cycle times, elevated contamination risk, and compounding product yield losses of 5–10% per cycle. SaniSure’s latest Mixed4Sure™ & One2Fill™ Application Note presents a validated, end-to-end single-use architecture that eliminates these bottlenecks entirely:
Across the full integrated workflow, processing time is reduced from 185 minutes to under 17 minutes per cycle — a 90–94% efficiency gain — with product recovery exceeding 98.5% and hold-up volume below 1% of batch volume.
Ready to eliminate intermediate transfers and compress batch cycle times? Access the complete Application Note — including full workflow diagrams, quantified ROI data, and assembly specifications.
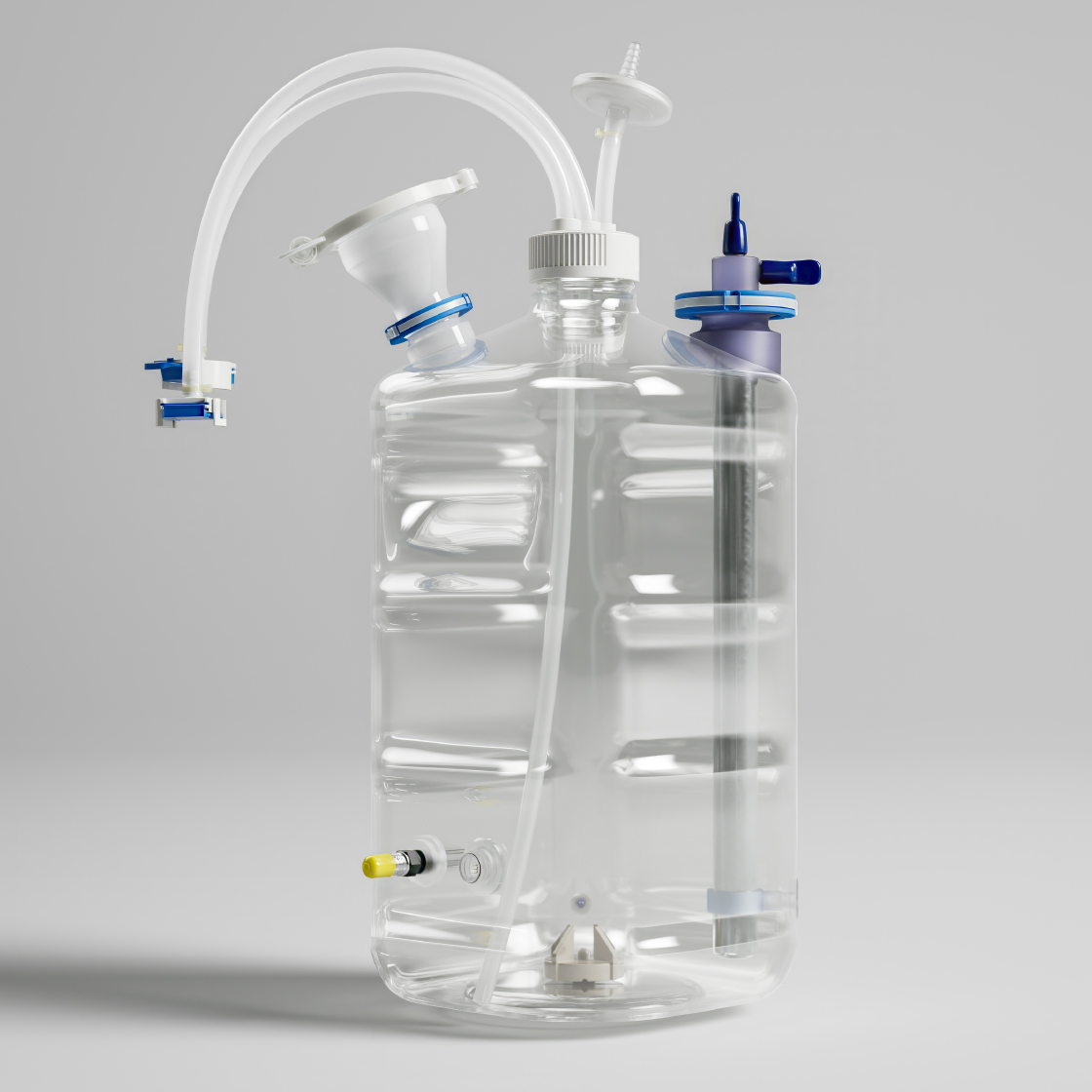
Our Comprehensive Workflow Solution
Most workflows treat mixing and filling as separate challenges. Mixed4Sure™ and One2Fill™ are designed to work together as a single closed system, from first mix to final fill.