Eliminate manual transfers, reduce contamination risk, and compress batch cycle times with a closed, single-use workflow for mixing through sterile aliquoting.
The News & Events
Explore how bottom-mounted mixing improves product integrity, boosts operational efficiency, increases mechanical reliability, and supports regulatory compliance.
Modernize vaccine and viral vector manufacturing with automated roller bottle processing that reduces contamination risk while meeting FDA and EU regulatory requirements.
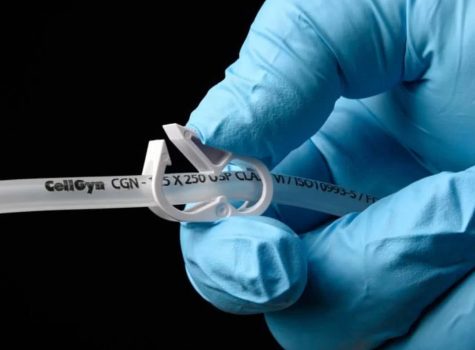
TR-1092 study proves ClearGreen®Pure outperforms leading PVC tubing in weld integrity, burst pressure, and USP <788> compliance for bioprocessing applications.
Strengthen bioprocessing supply chains with SaniSure’s USA and EMEA based manufacturing. Eliminate import delays, reduce hidden costs, ensure FDA compliance.
Discover how SaniSure delivered 70 custom assemblies in half the standard lead time, supporting vital viral vector manufacturing for a leading CDMO.
This comprehensive white paper examines the critical performance characteristics of SaniSure’s® CellGyn® tubing in aseptic biopharmaceutical applications.
This white paper explores critical contamination control strategies and advanced containment solutions to protect sensitive mRNA and gene therapies in GMP biopharmaceutical manufacturing environments.
This study validates gamma irradiation sterilization methods for single-use bioprocessing systems, demonstrating effective sterility assurance using VDmax27.5 method.